Dordan specializes in the design of custom thermoformed packaging solutions, like medical packaging, clamshells, blisters, inserts, and dunnage trays.

Above: Thermoformed dunnage tray design that protects products and offers shipping and assembly efficiencies
We employ a risk based approach to engineering integrated with design for manufacturing principles to supply high quality packaging that meets the customer's requirements.
Dordan uses Siemens NX CAD for developing its thermoform packaging designs. Structured by the ISO quality management system, our engineering process is highly documented and thorough to ensure each potential failure scenario has been addressed before developing a design.
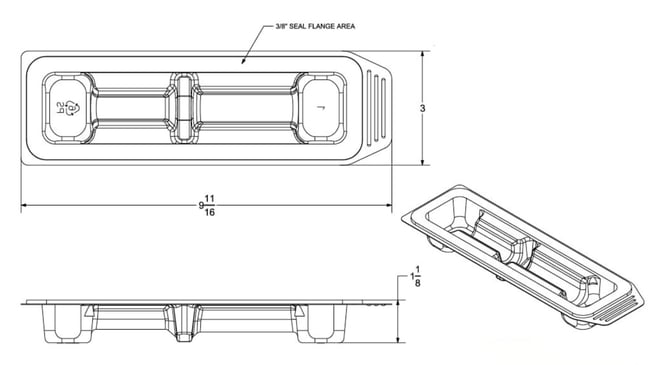
Above: Basic medical device tray engineering drawing
We provide:
Photorealistic Renderings
Engineering Drawings
3D Model Files
3D printed prototypes
Formal Prototyping
Risk-Based Engineering
Design-For-Manufacturability Evaluations
Final Production Drawing
Clamshells, Blisters, and Insert Trays: Secure, organize, and display consumer products. Designed for visibility, retail presentation, and impact resistance.
Dunnage, Shipping, & Assembly Trays: Durable trays engineered to protect automotive and industrial components. Ideal for shipping, WIP, and robotic assembly integration.
Medical Trays & Packaging: Designed to ensure sterility, compliance, and device integrity. Manufactured in or out of our ISO Class 8 cleanroom.

Above: Plastic clamshells showcase and protect product at retail
Our thermoform design process includes:
Cross-functional reviews from engineering, production, and quality control determine design risks, performance criteria, and quality control checkpoints.
3D printing and prototyping for rapid validation
After prototype approval, production tooling and components are machined using identical tool paths, minimizing translation errors and ensuring repeatability.
All tooling components, from assist plugs and trim dies to pressure boxes and EOAT, are built and assembled in-house.

Above: The same team of packaging engineers that develop the design also build the tooling
We specialize in custom medical thermoform package design for Class I, II, and III devices, offering solutions that support sterile barrier systems, aseptic presentation, and regulatory requirements.
Our medical packaging design services include:
Device-Specific Forming Cavities: Tailored to the geometry and handling needs of surgical tools, implants, and diagnostic components.
Aseptic Presentation Considerations: Trays are engineered for intuitive loading and access during sterile field transfer, supporting clinician usability.
Sterile Barrier System Integration: Designs accommodate Tyvek, film, or foil lidstock to create validated sterile packaging systems.
ISO 9001:2015 & ISO 13485:2016 Support: Our design process aligns with key regulatory standards and validation requirements.
Rapid Prototyping & Iteration: Fully integrated design and tooling services allow for quick design and prototype iterations.

Above: Medical kit packaging organizes, orients, and protects critical devices and components
What Sets Us Apart
63-Years of Thermoform Packaging Design
Fully-Integrated Packaging Designer & Manufacturer
Risk-Based Engineering & Design for Manufacturing
4th-Generation Family-Owned & Made In The USA Since 1962
Request a Design Consultation
Contact Us or Request a Quote for custom designed thermoformed packaging.
Phone 815-334-0087 | Fax 815-334-0089 | Toll Free 800-663-5460 | sales@dordan.com
2025 Castle Road, Woodstock, IL, 60098
©2018 Dordan Manufacturing Company, Incorporated. All Rights Reserved.