Dordan manufactures precision engineered medical packaging solutions like medical device trays and surgical kits. We cater to the medical and healthcare industry with ISO 13485:2016 certification, complete design through manufacturing process control, and ISO Class 8 cleanroom manufacturing.

Above: Surgical kit packaging protects products and maintains device sterility and efficacy
Thermoformed medical packaging is used to securely contain and protect medical devices throughout sterilization, shipping, storage, and point-of-use handling. These rigid plastic trays, kits, and blisters are custom-formed to fit the exact dimensions and functionality of the device, ensuring performance in both sterile and non-sterile environments.
Sterile Barrier Systems: Designed to be sealed with Tyvek, film, or foil lidstock, these trays maintain sterility until the point of use.
Device Protection: Thermoformed packaging is engineered to prevent movement, abrasion, and contamination, ensuring safe delivery from manufacturer to end user.
Ease of Use: Medical trays and packaging are custom-designed for intuitive loading and opening, supporting aseptic presentation in clinical settings.
Regulatory Compliance: Thermoformed packaging is validated to meet industry standards, supporting FDA and EU MDR requirements for medical device packaging.
Custom Fit and Functionality: Every tray is tailored to the shape, weight, and handling needs of the device it protects, optimizing both workflow and safety.

Above: Thermoformed medical clamshell packaging keeps clean critical medical devices and components
Thermoforming lends itself to medical packaging for its cleanliness, transparency, repeatability, and quality.
Medical Device Trays: Sealed to Tyvek or other lidding, medical device trays protect products and maintain device safety and efficacy.
Surgical & Procedural Kits: Organize and orient multiple medical components and facilitate operational workflows with custom medical kit packaging. Composed of multiple trays, thermoformed surgical kits are sealed to lidding or put in pouches to ensure sterility to the point of use.
Medical Clamshells: Completely contain your medical device in rigid plastic with medical clamshell packaging. Medical clamshells protect and orient medical devices and are often times put in pouches before sterilization.
Medical Blisters: Sealed to lidding, medical and pharmaceutical blisters maintain the cleanliness of healthcare products.
Medical Component Handling Trays: Optimize work in process, material handling, shipping, and assembly with medical component handling trays. Designed for product protection and high density, thermoformed handling trays safely move medical components throughout the supply chain.

Above: Medical component handling tray for efficient shipping and assembly
From concept development through cleanroom manufacturing, our vertically integrated model ensures speed, quality, and control.
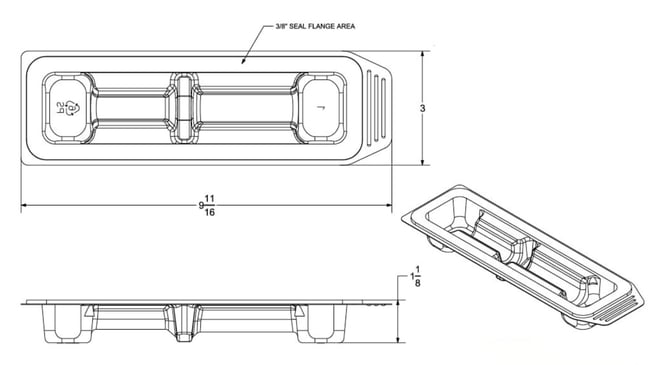
Above: Basic medical device tray engineering drawing
Our integrated capabilities include:
Medical Package Design: We use Siemens NX for our medical packaging designs. Employing a risk based and design for manufacturing approach, Dordan develops medical packaging that meets the functional, regulatory, and usability requirements of its customers.
In-House Tooling: NX CAD/CAM CNC machined tooling cut from a solid block of aluminum. All CNC tool paths are programmed by the same packaging engineers that developed the design, eliminating translation errors for more intelligent quality control. Once the prototype is approved, the geometry is replicated via step and repeat across the surface of the production tool so there are no variations between cavities.
ISO Class 8 Cleanroom Thermoforming: Dordan's fully enclosed and complete hardwall construction ISO Class 8 cleanroom supports the manufacture of sterile barrier packaging and contamination-sensitive devices. The cleanroom features temperature and humidity control, cascading airflow, particle count monitoring, and routine deep cleaning.
ISO-Certified Quality Management: Our operations are ISO 13485:2016 and ISO 9001:2015 certified, with process validation, inspection protocols, and traceability systems designed for FDA- and MDR-regulated packaging programs.
Our in-house, ISO Class 8 certified cleanroom is designed to meet the stringent requirements of the medical device industry, ensuring that every product we manufacture upholds the standards for cleanliness and quality.

Above: Dordan thermoforming medical packaging in its cleanroom
Cleanroom features:
Fully Enclosed Hardwall Construction: Our cleanroom is a 3,200 sq. ft. facility with hardwall construction, providing a controlled environment that minimizes particulate and microbial contamination .
Advanced Thermoforming Equipment: Equipped with Keifel Speedformers, our cleanroom facilitates precise forming, cutting, stripping, and stacking of medical packaging, ensuring consistent quality and performance.
Air Quality Management: We maintain a positive air pressure differential between the cleanroom and adjacent areas, coupled with cascading filtered airflow, to prevent the ingress of contaminants.
Regular Monitoring and Cleaning: Our cleanroom undergoes random particulate count testing and is subject to rigorous cleaning protocols, including pre- and post-production deep cleaning and sterilization via dry fogging.
Gowning and Material Handling Procedures: We enforce strict gowning procedures and have dedicated material entry rooms to further reduce the risk of contamination.
63-Years of Custom Thermoforming Experience
ISO 13485:2016 | ISO Class 8 Cleanroom
4th-Generation Family Owned and Operated
Made in the USA Since 1962
Phone 815-334-0087 | Fax 815-334-0089 | Toll Free 800-663-5460 | sales@dordan.com
2025 Castle Road, Woodstock, IL, 60098
©2018 Dordan Manufacturing Company, Incorporated. All Rights Reserved.