Dordan Manufacturing specializes in custom thermoformed medical device trays designed for sterility, performance, and ease of use. These trays are engineered, tooled, and manufactured in house, allowing for the seamless transition from tray concept to final part.

Above: Medical device tray protects and maintains the sterility and safety of medical devices
Thermoformed medical trays are rigid plastic packages sealed with Tyvek or other lidstock materials to create a sterile barrier system. They are easy to load, seal, and sterilize; resistant to punctures and contamination; and designed for intuitive opening and safe device removal. They are also preferred by practitioners for their durability and simplicity.
Medical device tray features:
Raised seal flange for Tyvek or lidstock application and clean, secure seals
Snap-fit, friction-fit, and press-fit cavities to hold devices in place
Finger access and peel tabs for intuitive and safe device removal
Pragmatic tag and die design for clean cut edges and minimal flash
Medical grade material PETG material for strong form and consistent sealing
For customers who prefer pouch packaging, trays can also be placed in Tyvek pouches as an alternative sterile barrier system.

Above: Medical device tray with raised seal flange offers intuitive opening and device removal
Dordan offers end-to-end medical device tray packaging solutions. From in house design to cleanroom manufacturing, Dordan maintains complete process control to ensure the production of high quality medical device trays.
Medical Packaging Design: Utilizing Siemens NX CAD solid modeling for developing medical trays, Dordan incorporates design for manufacturing and risk-based engineering for high quality production.
CNC Prototype and Production Tooling: NX CAD/CAM CNC integration allows for zero translation errors between tray design and prototype and production tooling. The same team of engineers that develop the packaging concept program the CNC tool paths, optimizing quality control and facilitating timeline gains.
Custom Thermoforming: Dordan's standard factory houses 14 in-line thin-gauge thermoforming machines. Appropriate for non critical medical and healthcare applications, we offer multiple tiers of quality control to ensure that the parts being produced meets the customer's performance requirements.
ISO Class 8 Cleanroom: A controlled environment ideal for sterile medical packaging applications. Monitored particulate, temperature and humidity, Dordan's cleanroom is a fully enclosed and complete hardwall space equipped with two Kiefel thermoforming machines.
ISO 9001:2015 and ISO 13485:2016 Certified: Dordan's quality management system strives to ensure the on time delivery of defect free medical trays and packaging.
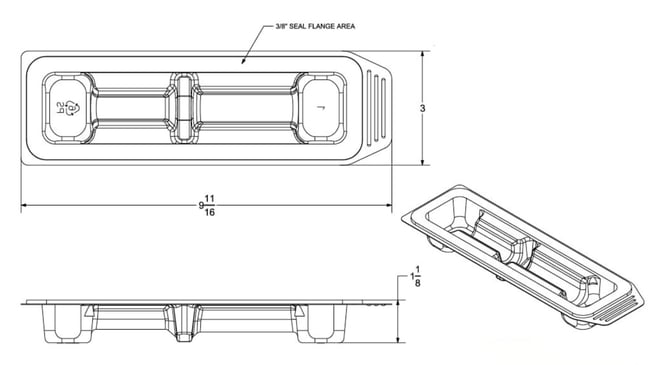
Above: Medical device tray engineering drawing
Dordan is equipped with an ISO Class 8 cleanroom for manufacturing medical device trays. This controlled environment is ideal for particulate-sensitive medical devices and components.
Cleanroom features include:
Kiefel Speedformers with automated forming, die cutting, stripping, and stacking
Cascading filtered airflow design to manage particulate and bioburden levels
Terminal cleaning protocols before and after each production run
Annual sterilization via antimicrobial dry fogging
Compliant gowning rooms and material staging zones for controlled entry
Particulate count testing and environmental monitoring performed regularly
Enhanced quality control inspection and documentation, including First Article Validation and CPK studies

Above: Dordan's cleanroom manufacturing medical device trays
Fully-Integrated US-based Medical Tray Packaging Supplier
In-House Prototyping & Tooling for Rapid Development
4th-Generation Family-Owned Since 1962
Risk-Based Engineering & Design for Manufacturing
Contact Us today or Request a Quote to get started on your medical packaging project.
Phone 815-334-0087 | Fax 815-334-0089 | Toll Free 800-663-5460 | sales@dordan.com
2025 Castle Road, Woodstock, IL, 60098
©2018 Dordan Manufacturing Company, Incorporated. All Rights Reserved.