Medical device trays protect and keep clean medical devices. When sealed to Tyvek lidding and sterilized, medical trays and blisters maintain the safety and efficacy of the device. Here are the main design considerations when developing medical device packaging.
Components required for medical tray packaging
To make medical device tray and blister packaging that is sealed to lidding, one must secure a heat sealer (or outsource), a thermoform supplier, a lidding supplier, or a one-stop-shop. Custom heat seal tooling is required to seal medical blisters and trays to lids, which can be developed by the equipment supplier. If the sealed package is to be sterilized, one must determine the appropriate sterilization method and enlist the corresponding partner.
It is advised that the packaging development process begin with the thermoformed tray (not the lidding or heat seal tooling). This is because the lidding and heat seal tooling need to account for tolerances inherent in the thermoforming process, which won't be determined until the medical blister has been fully engineered (or produced).
Medical tray and blister manufacturing environment
Medical device trays and blisters can be manufactured in a certified cleanroom or standard production facility. The goal of a cleanroom is to monitor, manage, and mitigate the presence of particulate (dust, fibers, skin cells), thereby reducing the potential for bioburden agents. This is accomplished with robust air filters and a cascading air flow design married with strict personnel compliance to all cleanroom SOPs.
Most medical products that are to be packed and sealed in a cleanroom require that the medical packaging also be produced in a cleanroom; many medical products that are going to be sterilized also require cleanroom-produced thermoformed packaging.
It is not just the cleanliness of cleanrooms that dictate if a medical blister should be manufactured in it or not; it is also the level of quality control. Equipment in cleanrooms tend to be more sophisticated then standard production facilities, facilitating more control over the thermoforming process to achieve and maintain tighter tolerances.
Lastly, with cleanroom production, process capabilities studies are conducted utilizing automated optical inspection equipment to generate reports for customers' review and approval prior to beginning high-volume production.
Seal Flange
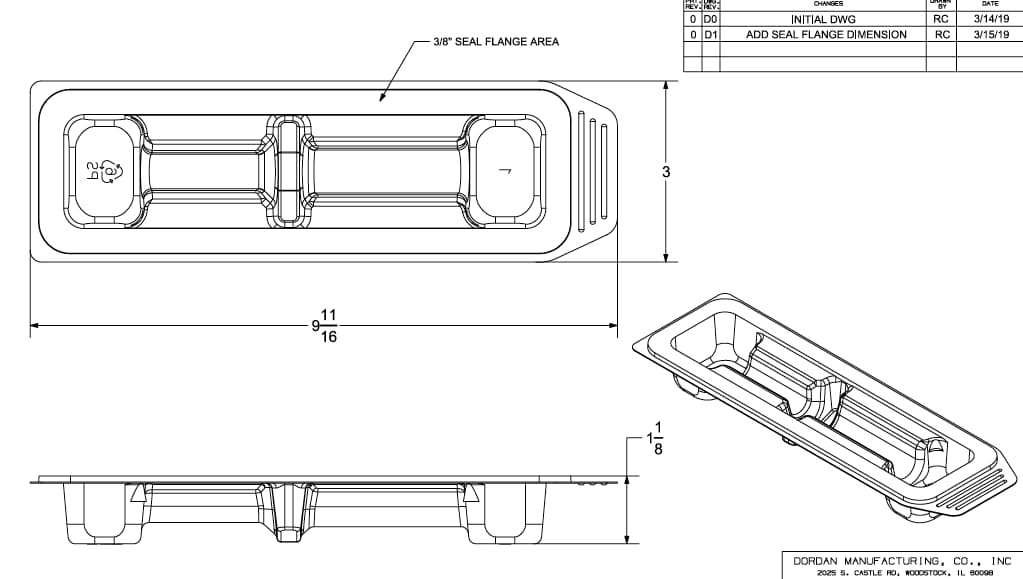
The seal flange is an extremely important part of the medical device tray and blister design, especially when the packaging is going to be sterilized. If the seal between the Tyvek and tray or blister is broken, then the product is no longer sterile. There are two kinds of seal flanges for medical trays and blisters: standard seal flanges and raised seal flanges.
Standard flange on a medical device tray or blister is the space between where the steel rule die cuts the thermoformed part from the plastic web and the part sidewall. To achieve a good seal, the medical blister/tray flange must maintain a consistent material thickness to mitigate the potential for any voids in the seal. The strength of a seal is based on maintaining the validated heat sealing parameters and using high quality heat seal tooling, thermoformed trays, and lids that are all developed in tandem.
Sealing blisters with standard flanges to lidding is popular for some small and lightweight medical products. If you wish to seal to the medical blister's flange, it is encouraged that the flange be at minimum, 0.25". Draw backs to medical blisters with standard seal flanges are limitations regarding peel tab design and the potential for less structure of the seal flange.
Raised seal flanges are essentially formed bumpers around the medical tray cavity that allow for a robust seal and offer enhanced product protection and medical packaging stability. The material distribution on the raised seal flange is more consistent then with standard flanges because it is a formed feature, not a flat piece of die cut plastic. Medical trays and blisters with raised seal flanges tend to be favored over standard seal flanges by many medical device OEMs.
The image below is a medical device tray with raised seal flange, followed by an engineering drawing.

Material thickness and seal
The thickness of the medical device tray or blister material starting gauge needs to be determined by the weight of the product, the depth of draw, and the interior cavities. While the starting gauge doesn't have a direct correlation to the strength of the seal, there must be enough material to draw from to allow for a consistent material distribution throughout the thermoformed blister. If the material is too thin and is stretch too far over and into the tooling, there is the potential for pin holes or voids on the seal.
Voids result when there is inconsistency in material thickness on the seal flange. Here, the thicker parts of the blister or tray flange are sealed first and the difference in thicknesses can lead to voids depending on the variation in the sealed area thickness.
Material
Medical trays and blisters are typically produced in Medical Grade PETG: It is the best material for manufacturing medical packaging because the ‘G’ reduces the brittleness of the part (when compared to PET) and facilities a good seal. Medical PETG is produced in ISO 9001/ ISO 13485 facilities, where care is taken to maintain the specified quality requirements. Medical Grade PET is also produced with this level of quality control; however, because the material minimums are so high in comparison to Medical PETG, it isn't often times the most economical choice. FDA PET is not medical grade and therefore doesn’t have the level of cleanliness required to be brought into many cleanrooms. PET/RPET (recycled PET) is the same material that water bottles are made from and can be used for some healthcare packaging, depending on the quality requirements. FDA HIPS isn't clear (white, black), but can be used for applications that don't require transparency.
Silicone
Silicone is a coating that is applied to plastic sheet after the extrusion process to facilitate denesting of the thermoformed parts (and keep the cored plastic from sticking to itself when being fed into the thermoforming machine). PETG is available as single sided silicone or double sided. Many medical devices customers do not want the product to come in contact with silicone, so single sided PETG is required. Here, it is important to have the silicone-side of the plastic sheet mold side (underbelly of tray or blister).
For medical products that are contaminated by the presence of any silicone, medical grade materials with internal denests are available. There are also other materials for PETG that are not silicone-based, but do have the slippage requirement.
Medical device tray packaging is an efficient way to safely bring medical products to market. If you enlist high quality packaging suppliers, the development of the medical blister packaging will be managed by a team committed to the production of problem free medical blister packaging.

